- How Blue Light Impacts Your Sleep Quality
- Comparing Whey and Plant-Based Protein: Which is Best?
- How Long Does Nicotine Remain in Your System?
- The Best Time of Day to Drink Bone Broth to Maximize Health Benefits
- 8 Ways to Increase Dopamine Naturally
- 7 Best Breads for Maintaining Stable Blood Sugar
- Gelatin vs. Collagen: Which is Best for Skin, Nails, and Joints?
- The Long-Term Effects of Daily Turmeric Supplements on Liver Health
- Could Your Grocery Store Meat Be Causing Recurring UTIs?
- Are You Making This Expensive Thermostat Error This Winter?
New Test Estimates Risk of Heart Disease
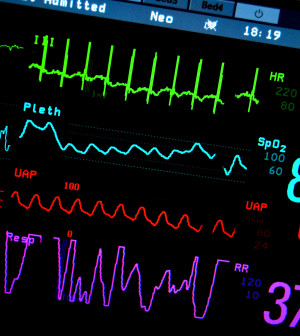

A new screening test designed to estimate a person’s risk of developing coronary heart disease (CHD) has been approved by the U.S. Food and Drug Administration.
Results of a manufacturer study reviewed by the FDA indicate the test is better at discerning this risk in women, notably black women, the agency said in a news release.
The PLAC Test for Lp-PLA2 Activity, intended for people without a prior history of heart disease, measures a blood biomarker for arterial inflammation, a strong indicator of plaque buildup within arteries that supply blood to the heart.
Heart disease is the leading cause of death in the United States among ethnic groups including blacks, Hispanics and whites. The most common form of heart disease, CHD, leads to the deaths of some 385,000 people each year. About two-thirds of women and half of men who die suddenly of CHD have no prior symptoms, the FDA said.
The new test is produced by diaDexus Inc., based in South San Francisco.
More information
Visit the FDA to learn more.
Source: HealthDay
Copyright © 2026 HealthDay. All rights reserved.










