- How Blue Light Impacts Your Sleep Quality
- Comparing Whey and Plant-Based Protein: Which is Best?
- How Long Does Nicotine Remain in Your System?
- The Best Time of Day to Drink Bone Broth to Maximize Health Benefits
- 8 Ways to Increase Dopamine Naturally
- 7 Best Breads for Maintaining Stable Blood Sugar
- Gelatin vs. Collagen: Which is Best for Skin, Nails, and Joints?
- The Long-Term Effects of Daily Turmeric Supplements on Liver Health
- Could Your Grocery Store Meat Be Causing Recurring UTIs?
- Are You Making This Expensive Thermostat Error This Winter?
Sealant Gel Approved for Eye Surgery
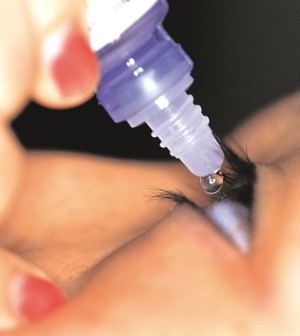

A sealant gel to prevent fluid leakage after cataract surgery has been approved by the U.S. Food and Drug Administration.
While gels such as ReSure have been approved to seal small incisions in other parts of the body, this is the first such approval for the eye, the agency said in a news release.
A cataract is a gradual clouding of the eye’s lens. By age 80, more than half of Americans have either an active cataract or have had cataract surgery, the U.S. National Institutes of Health estimates.
The ReSure Sealant kit includes two liquid solutions that are mixed together just before use. The product is designed to seal an eye incision within 20 seconds of application, the FDA said. The gel then breaks down over seven days and is naturally flushed away by the eye’s tears.
No serious adverse reactions to the gel were reported, the FDA said.
Before this approval, stitches were the only option for closing a leaking corneal incision after cataract surgery, the agency said. The product was evaluated in a clinical study of 471 adults.
The ReSure sealant is produced by Ocular Therapeutix, based in Bedford, Mass.
More information
The U.S. National Library of Medicine has more about cataracts.
Source: HealthDay
Copyright © 2026 HealthDay. All rights reserved.










