- How Blue Light Impacts Your Sleep Quality
- Comparing Whey and Plant-Based Protein: Which is Best?
- How Long Does Nicotine Remain in Your System?
- The Best Time of Day to Drink Bone Broth to Maximize Health Benefits
- 8 Ways to Increase Dopamine Naturally
- 7 Best Breads for Maintaining Stable Blood Sugar
- Gelatin vs. Collagen: Which is Best for Skin, Nails, and Joints?
- The Long-Term Effects of Daily Turmeric Supplements on Liver Health
- Could Your Grocery Store Meat Be Causing Recurring UTIs?
- Are You Making This Expensive Thermostat Error This Winter?
FDA Approves New Treatment for People With COPD

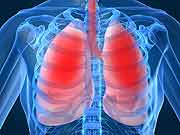
Patients with the progressive, deadly respiratory ailment known as chronic obstructive pulmonary disease (COPD) have a new weapon to battle the disease, the U.S. Food and Drug Administration said Thursday.
The agency approved a new treatment, an inhaled spray called Striverdi Respimat (olodaterol) for COPD, the third leading killer of Americans. COPD, which is often linked to smoking, involves multiple lung conditions such as chronic bronchitis and emphysema.
Patients with COPD may experience a number of symptoms, such as wheezing, coughing and tightness in their chest. According to the FDA, Striverdi Respimat belongs to a class of medicines known as long-acting beta-adrenergic agonists (LABAs).
The agency said the drug works by helping the airway muscles in the lungs stay relaxed, and it can be taken once daily over a prolonged period of time.
“The availability of this new long-term maintenance medication provides an additional treatment options for the millions of Americans who suffer with COPD,” Dr. Curtis Rosebraugh, director of the Office of Drug Evaluation II in the FDA’s Center for Drug Evaluation and Research, said in an agency news release.
The FDA evaluated the safety and effectiveness of Striverdi Respimat compared to a placebo in more than 3,100 people diagnosed with COPD. Those who took the medication had improved lung function.
Commonly reported side effects of Striverdi Respimat include runny nose, coughs, dizziness and upper respiratory tract infections, among others.
Striverdi Respimat is not approved for people with asthma, and asthma patients should not use it, the FDA warned. That’s because the drug raises the risk of asthma-related death. This inhalation spray should also never be used to treat breathing problems that develop suddenly, the agency cautioned.
People with COPD that is progressing rapidly should also not take Striverdi Respimat, the FDA added. For these patients, the medication could lead to heart problems and the narrowing or blockage of the airway, they explained.
More information
The U.S. National Heart, Lung, and Blood Institute provides more information on COPD treatments.
Source: HealthDay
Copyright © 2026 HealthDay. All rights reserved.










